
Reverse osmosis water is an incredible starting point for any aquarium owner seeking to create ideal water conditions for their fish and coral. RO water is completely free from chemicals toxic to fish like chlorine and chloramines, is neutral in pH, and contains no water hardness. Many tropical fish require specific water environments to thrive. Reverse osmosis allows you to control the parameters of your aquarium water’s chemistry, meaning you can support whatever aquatic life you desire. Saltwater and reef aquariums are notoriously difficult to maintain. Reverse osmosis simplifies the process by giving you a reliable, consistent pure water to craft your saltwater environment. In this article, you can discover answers to questions about using reverse osmosis for both freshwater and saltwater aquariums.
Is reverse osmosis water good for aquariums?Reverse osmosis water is ideal for aquariums because it produces a blank slate that you can manipulate to best suit your fish tank’s needs. RO systems produce high-purity water free of water hardness, chemicals like chlorine, toxins, and up to 99% of dissolved solids in the water. This provides a neutral environment for you to create the ideal conditions for your fish to thrive in. Reverse osmosis water allows you to design the precise water conditions that best suit your aquarium, unrestricted by your local water quality. Reverse osmosis also eliminates heavy metals, nitrates, salts, and dissolved minerals from water, leaving behind incomparable purity from which you can craft your aquarium environment. Reverse osmosis gives you total control over the pH levels of the water, the salt content, and what minerals your aquatic life will be exposed to.
If you have ever maintained an aquarium, you are intimately familiar with the importance of stable and predictable water quality. Even slight imbalances can precipitate illness and trigger declines in the health of your reef. Every fish requires a unique combination of pH and minerals to facilitate a happy and healthy life. South American cichlids like Red Oscars and Green Terrors thrive in very soft water that rests at a neutral pH. If your home uses well water or you live in regions with hard water, your water will not be able to support these magnificent, aggressive fish. African cichlids from Lake Malawi need very hard water with an alkaline pH of 8 or greater. These conditions mimic the waters in which these exotic fish originated before being disseminated as popular pets. It is very easy to imitate these conditions when you have pure water that will respond predictably to whatever minerals and pH buffers you introduce to the water.
Why is reverse osmosis so effective for aquariums?Reverse osmosis purifies water when pressure forces it through a semipermeable membrane covered in minuscule pores. The membrane allows water molecules to pass through its lattice of tiny pores, but contaminants are rejected. All salts, nitrates, phosphates, and minerals are unable to pass through the membrane and are drained out of the system. This waste, called the brine, flows from the reverse osmosis system to a drain. The purified water, called the permeate, flows out of the purification system and into a water storage tank.
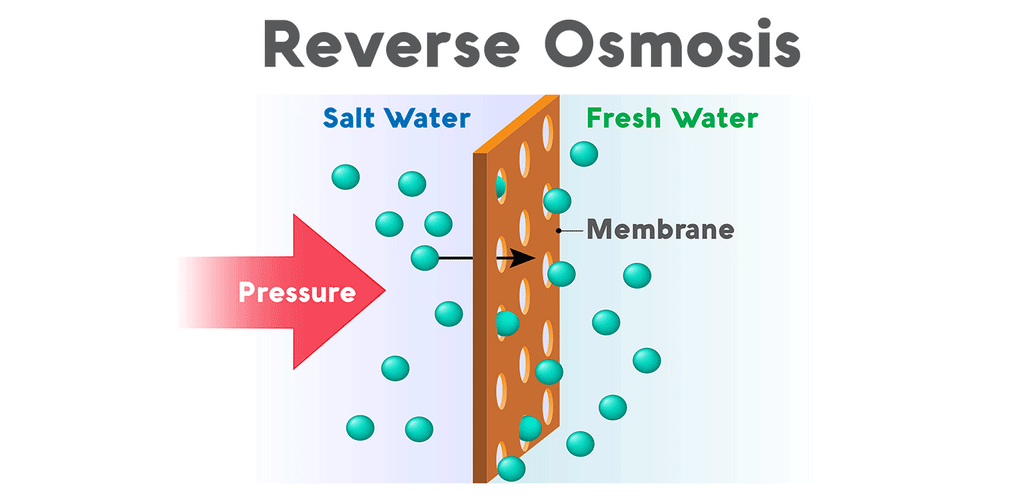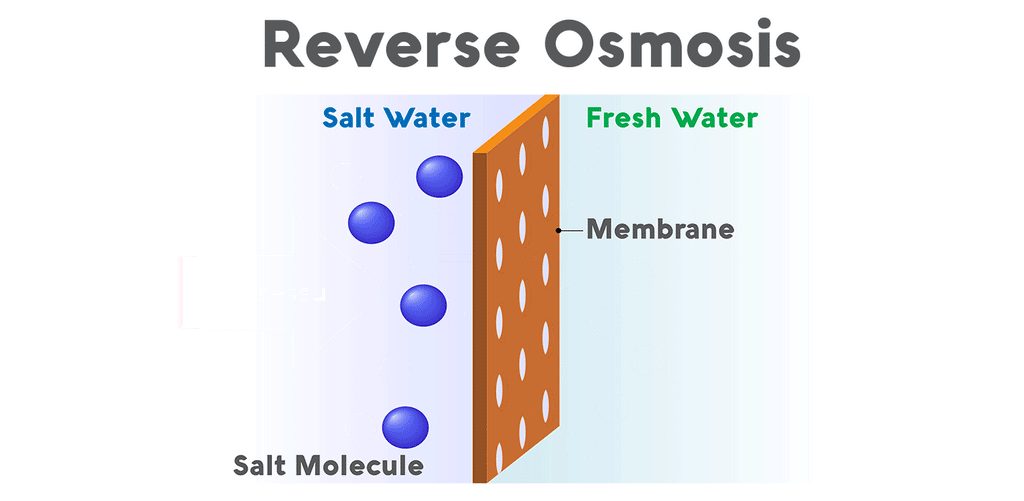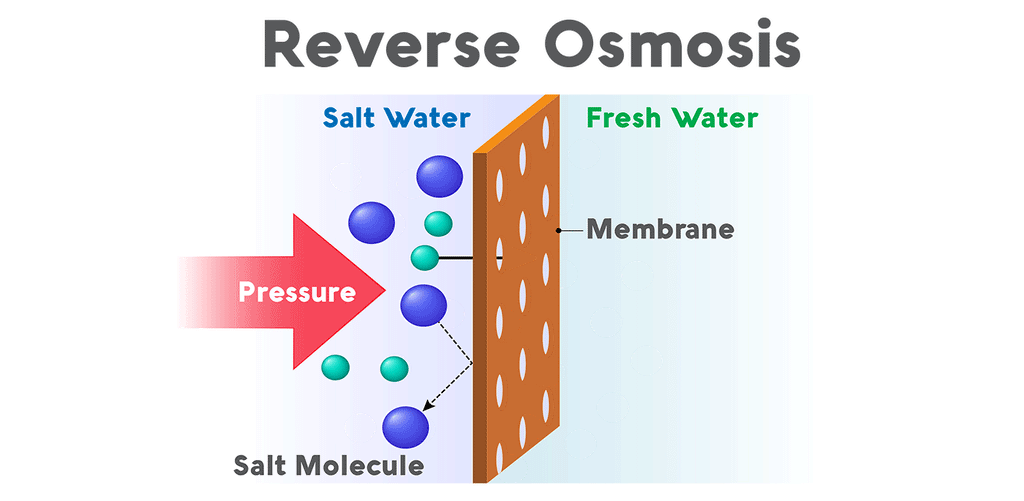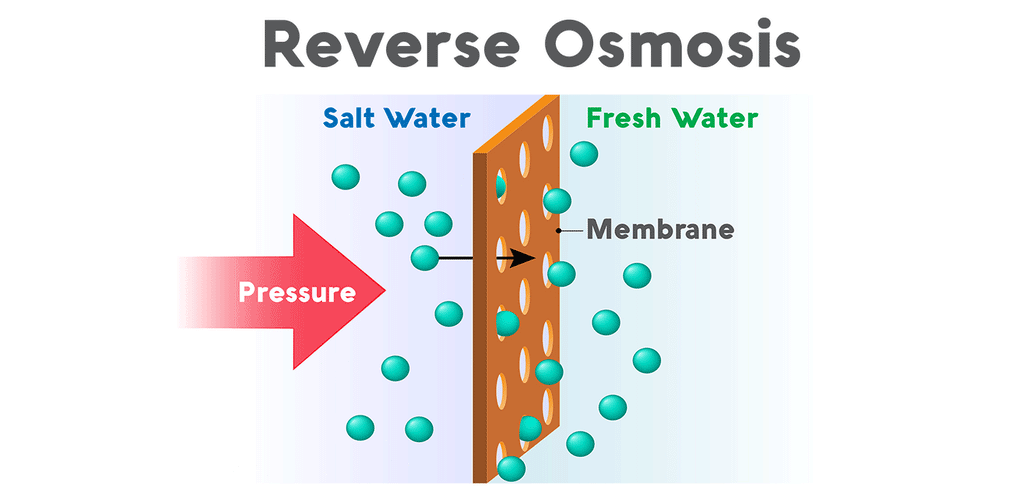
Reverse osmosis systems (also called RO) vary in the number of stages they use to process water. All reverse osmosis systems use a membrane to tackle the most thorough stage of filtration. However, reverse osmosis units also use a sediment pre-filter to eliminate any dirt, debris, or rust particles from the water. Though the membrane is very capable of removing sediment, too much of it can clog the RO system’s flow restrictor and cause the system to lose pressure. Most reverse osmosis systems also come equipped with both a carbon pre-filter and post-filter. Carbon removes chlorine and volatile organics from the water by a process called adsorption. This is a vital stage, not only because chlorine is very toxic to fish, but also because RO membranes are sensitive to chlorine. The thin-film composite membrane has a very low tolerance for chlorine, so a carbon pre-filter eliminates the chemical before contact. These pre-filters extend the lifespan of the membrane and boost the efficacy of the system.
Some RO units also have additional post-filters. Remineralizing cartridges can be added for those who want to boost the alkalinity of their water after the reverse osmosis process eliminates all minerals. This could be an ideal solution for those with aquariums that demand more alkaline pH levels. A deionization cartridge is also very popular among aquarists. Deionization, commonly called DI, further purifies water by trading any remaining ions in the water with hydrogen and hydroxide ions. Reverse osmosis systems combined with DI are often called RODI or RO/DI systems and are extremely popular choices for aquarium owners. The combination of RO and DI produces water of such absolute purity that it is sometimes used in laboratory, medical, and electrical applications.

What does a reverse osmosis system remove?
Reverse osmosis systems reduce the levels of fluoride, arsenic, benzene, calcium, magnesium, heavy metals like lead and copper, and pesticides in addition to many other dissolved organic and inorganic solids. However, there are specific compounds RO strips from water that makes it especially helpful for aquarium owners.
Nitrates
Nitrates, compounds commonly found in water supplies, are generated when nitrogen combines with oxygen. However, while humans have a tolerance to reasonable levels of nitrates, fish are much more sensitive to nitrogen compounds. As nitrate levels climb, algae will spread throughout the tank. Nitrates also suppress the oxygen levels in the water, which can negatively affect the wellbeing of all fish. If the levels reach over 100 parts-per-million(ppm), even hearty fish can become stressed and worsen in health. They can be especially dangerous to young fish and fry.
While freshwater aquariums can tolerate up to 40ppm of nitrates, reef aquariums are far less forgiving. Even slight nitrate levels can damage your corals and invertebrates. Sea cucumbers, such as the Sea Apple and the Tiger Tail, are extremely sensitive to nitrates and unable to handle exposure to them. Anemones, crustaceans, and other marine life will become ill at just 0.25 ppm. Though nitrates will be produced by fish as a waste byproduct, reverse osmosis ensures the starting level of nitrates is minimal. Nitrates can further be controlled in a tank by regular and diligent cleaning, mangrove plants, and natural nitrate reduction methods like using live sand and live rock with nitrifying bacteria.
Phosphates
Phosphates cause rapid algae growth within tanks. If left unchecked, this can lead to murky, green water and can decrease oxygen within the water. Though phosphates are not directly harmful to your fish, an aquarium overrun by algae will create health problems for its inhabitants over time. In reef aquariums, aggressive algae growth will interfere with healthy coral growth. Phosphates will spread across coral reefs, masking their vibrant colors and turning them a dull brown. Anything surpassing 0.2ppm can turn a reef aquarium into an algae field.
Though phosphates can be introduced into the tank by uneaten food, fish waste, and decaying plants, using untreated water will almost certainly raise your phosphate levels. It is common for tap water to have around 1ppm of phosphate in it, although some may have as high as 5-10ppm. Reverse osmosis eliminates phosphates from the water, guaranteeing you a clean start. Since you will have to control phosphate proliferation in your aquarium as part of maintenance, it is ideal to start with the lowest possible level of phosphate.
Silicates
As rainwater moves through rock deposits towards underground aquifers, it collects a mineral called silicate. Silicates are present in about 90% of the earth’s crust, making them one of the most common water contaminants. If you are using a sandy substrate in your aquarium, the silicates will leach out of the substrate into the water in a matter of weeks. Since silicates are so prevalent in water, the algae they produce is a rampant problem for many tank owners. Silica algae, commonly known as “gravel algae” spread across the rock bed before coating your entire aquarium in a brown film. The algae growth is further exacerbated by poor lighting and low oxygen levels in the aquarium.
Silicates can also be very dangerous to coral growth. Diatoms, a large family of single-celled algae, need silicates to grow. Some diatoms have a carnivorous effect on coral. Encrusting diatoms populate at the base of corals before reaching upwards. These diatoms will proceed to consume the coral, leading to the coral’s skeleton receding and decay and disintegration setting in. These diatoms can overtake healthy coral in days. Once again, reverse osmosis is the simplest way to eliminate this danger from your tank. Silicates cannot make their way past the RO membrane, reducing the risk of them emerging amongst your reef and gravel.
Water hardness
Hard water is water with elevated levels of calcium and magnesium that’s been accumulated during nature’s hydrologic cycle. Hard water is notorious for leaving crusty scale build-ups on faucets and showerheads, staining bathtubs and toilet bowls, and leaving behind streaky soap stains. Since it is not a health concern, water hardness is not regulated by municipalities, meaning many homeowners across the United States live in homes with very hard water.
Hard water limits the fish you can host in your aquarium. While there are many fish that thrive in hard water environments, such Swordtails, Guppies, and brackish fish like Archers, some freshwater fish have different temperaments. Tropical fish from the Amazon Basin, like Discus and Angelfish, are unable to thrive in anything other than soft water. Reverse osmosis makes water neutral and soft, allowing you to control the specific degree of hardness you desire (referred to by aquarists as General Hardness, or “GH”). This expands the range of choices available to you and your aquarium.

Reverse osmosis gives you a starting point to create the perfect marine environment for your saltwater fish. A successful saltwater aquarium relies heavily on a strong foundation. Even trace amounts of compounds can throw off the entire composition of your reef tank. For example, even low levels of nitrates can cause algae to bloom and proliferate throughout your saltwater aquarium. Maintaining the delicate balance of water parameters is integral to the success of the tank. Reverse osmosis systems provide you with water you have total control over.
Since very few people have access to the actual seawater conducive to their marine life, aquarists have found starting with total water purity is the easiest way to build their water profile. Tap water contains disinfection byproducts and minerals unnatural to seawater environments. Mixing sea salts with tap water can get you an approximation of the ocean’s water, but it cannot replicate it. Reverse osmosis water provides consistent water quality. You do not need to perform elaborate water tests and expend significant effort working around your water quality to create the water you desire. You know you are starting with slightly soft water devoid of contaminants and chemicals. From here, you can increase the alkalinity and hardness and add the desired levels of salt. The power to create the ideal water conditions rests in your hands. Reverse osmosis brings welcome consistency and predictability to a hobby notorious for its difficulty.
What are the best reverse osmosis systems for aquariums?The best reverse osmosis system for your aquarium depends on your desired level of purity, what kind of tank environment you are seeking to develop, and the size of your fish tank. If you are maintaining a large aquarium, you will want a system with a high output rate. RO membranes are rated by the number of gallons of water per day that they can output. A high-output home reverse osmosis system and large RO tank will prevent you from constantly waiting for water to fill your aquarium with. The reverse osmosis process works quite slowly, and you can’t afford to wait around for water to refill your tank. The Neo-Pure PRO-4 75 GPD RO System and the Pentair Freshpoint GRO-475M provide a high-output rate for a residential RO system, enough to keep up with constant topping off of a small to medium-sized fish tank. The Neo-Pure system also includes a permeate pump. These reduce up to 85% of the waste generated by reverse osmosis, enhance the performance of the membrane, and increase the pressure and volume of the RO storage tank.
If you want to achieve the maximum water purity, add a DI cartridge between your carbon post-filter and storage tank to further reduce the TDS to zero. Reverse osmosis combined with deionization is the preferred choice by many aquarists. RODI produces water that is around 99% pure water. Reverse osmosis systems will usually remove around 95-98% of all total dissolved solids. Deionization uses ion exchange to capture and eliminate whatever slight particulate matter might have escaped the RO membrane. The result is water of incredible clarity and purity. RODI systems are especially useful for saltwater aquariums and reef applications. Corals and marine invertebrates are very delicate and quite sensitive to any shift in the water. To preserve their vitality and foster growth, RODI systems are used. They produce water of reliable and consistent quality that will not disturb these creatures. Some marine fish also have very specific parameters required for their survival. RODI assures that you have maximum control over creating their environment and don’t risk harming your creatures.
How do I adjust my reverse osmosis aquarium water?Once you’ve obtained the contaminant and mineral-free water produced by reverse osmosis, you are now able to regulate the water profile to best match your fish’s needs. Three of the most important parameters of aquarium water chemistry are general hardness (GH), carbonate hardness (KH), and the pH balance of the water. In general, most fish will favor either a softer, acidic environment or harder and more alkaline waters.
Why is general hardness (GH) important in an aquarium?
The general hardness of your aquarium water is the measurement of your water’s calcium and magnesium content. The more minerals present in the water, the harder the water will be. As water journeys through the earth’s layers toward underground aquifers, it passes through rocks like gypsum and limestone. Water, a universal solvent, will dissolve minerals from these calcium and magnesium-rich rocks. The hardness of your municipal or well water supply will vary depending on the regional make-up of the earth’s crust.
In general, most fish do have a GH preference reflecting their natural environments. While many fish can tolerate some degree of hardness, it becomes extremely important for breeding. Many fish will not reproduce in a climate dissimilar to their natural habitat. Furthermore, general hardness is integral to a process called osmoregulation. Osmoregulation is the process by which fish regulate the salt concentration and water inside their bodies with the salt concentration and water they are immersed in. If the water is too hard or too soft, the fish’s internal balance will be thrown and the fish can perish. The GH of the water is also where fish will obtain the electrolytes they need to encourage bone and muscle growth, strengthen immunity, and overall development. Magnesium aids in the fish’s ability to digest food.
A fish’s preferred GH varies wildly species to species. Many saltwater systems will have a general hardness as low as 2-3 degrees (0-50ppm) because ocean water has very low exposure to limestone rocks. East Asian paradise fish prefer harder water, and African cichlids do best in water as high as 20 dGH (over 300ppm).
| Degrees | PPM (Parts-Per-Million) | Water Hardness |
| 0 - 4 dH | 0 - 70 ppm | Very soft |
| 4 - 8 dH | 70 - 140 ppm | Soft |
| 8 - 12 dH | 140 - 210 ppm | Medium |
| 12 - 18 dH | 210 - 320 ppm | Mildly hard |
| 18 - 30 dH | 320 - 530 ppm | Hard |
| 30+ dH | 530+ ppm | Very hard |
How to adjust general hardness (GH) in your aquarium
To lower your aquarium’s general hardness, your best choice is to start with reverse osmosis water. Removing dissolved mineral content is a very difficult task, and if your water contains any significant degree of hardness, you will be unable to do so without filtration. This is another reason why reverse osmosis is so prevalent in saltwater aquariums. The low GH demanded by these tanks is best achieved by using RO water or by mixing in distilled water. Adding peat moss, plant substrate, and driftwood to your tank will also absorb some of the mineral content, but it will also bring down your water’s pH level.
To increase your aquarium’s general hardness, add mineral-rich limestone to the tank. Crushed coral and aragonite can be mixed into your substrate. These are made of calcium carbonate and will increase the general hardness of your tank as they dissolve. However, these are best paired with fish who prefer high KH and pH, like cichlids, as they tend to raise those levels as well as GH. Remineralizing buffers are also sold that can be added to the water to elevate GH levels.
Why is carbonate hardness (KH) important in an aquarium?
Carbonate hardness is the measurement of carbonates and bicarbonates present in your water. KH functions as a buffer to your aquarium’s pH. Most fish, coral, and aquatic plants react very poorly to any rapid pH fluctuations. Changes in pH shock fish and even results in their death. KH, also sometimes referred to as your tank’s alkalinity, ensures that acids do not immediately attack the pH balance of the tank. Instead, any change to pH is first softened by the KH of the water.
As your fish create waste, ammonia and nitrites are natural byproducts. These are acidic compounds, and without a buffer between them and the pH, they would increase the acidity of your water. However, the acids instead eat away at the aquarium’s KH. In saltwater tanks, the bicarbonates also help boost the growth of the coral’s exoskeletons. Out of all the water chemistry parameters of the tank, KH arguably plays the most important role. Without stable KH, you risk your pH crashing. KH should never reach zero because this would eliminate any buffer between the pH and instability.
Different fish do have different KH preferences, just like they have for hardness and pH. Normally, the higher the pH of your water, the higher levels of KH will be required. Most fish and plants are comfortable in a relatively large range of KH (anywhere from 1-10 dKH). Monitoring your KH to ensure it is staying balanced is the most important factor. It should not be adjusted unless necessary.
How to adjust calcium hardness (KH) in your aquarium
To lower your KH, there is nothing more effective than reverse osmosis water. Starting with RO/DI water will give you water totally devoid of KH, and therefore allowing you to raise the KH to whatever levels are best suited for your fish and your pH. Natural additions like Indian almond leaf and peat moss can gently lower the KH of your aquarium. You can also use an acid buffer, which will convert calcium carbonate and bicarbonate into carbon dioxide. However, be very cautious with acid buffers. These are best used in tanks where plants absorb carbon dioxide, as this can cause your KH and pH to plummet and result in serious harm to your fish.
To increase your KH, you can use sodium carbonate or bicarbonate (more commonly called soda ash and baking soda). These will introduce more carbonate into your water and boost the water’s KH. Aquarium stores also sell alkalinity buffers that come with guides on how much to add based on your aquarium requirements. Natural media like dolomite rocks, aragonite, and crushed coral will all also raise KH. However, keep in mind, these also will impact your water’s GH and pH levels.

Why is pH important in an aquarium?
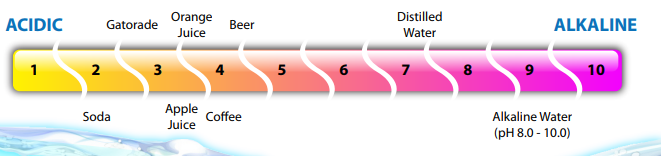
Broadly speaking, pH is a measurement of how acidic or alkaline your aquarium water is. pH is measured on a scale from 1 to 14, with the middle number 7 representing a neutral value. Anything below 7 is considered to be acidic, while any value above 7 is considered to be basic (or alkaline). The pH scale is logarithmic, which means each number increase or decrease is ten times greater than the number preceding it. For example, a pH reading of 8.5 is ten times more alkaline than a reading of 7.5. However, a pH of 9.5 would be one hundred times more alkaline than 7.5. This is why dramatic changes in pH are so dangerous. A drop of several points means the water is now thousands of times more acidic than before.
Historically speaking, pH was the most emphasized aquarium water chemistry parameter. This was both because it is simple to measure the pH of water and because of a more limited understanding of how aquarium water chemistry worked. However, we now understand that the calcium hardness (KH) of the water has a lot more influence over the acidity or alkalinity of the water than the pH. This is because the water’s pH can change without affecting the water’s KH. However, if the water’s KH drops, the water’s pH will always be affected. KH is the buffer to protect the fish from the changes in pH. The pH itself can shift a few points without hurting the fish and coral if the KH is holding stable.
In general, fish that prefer softer water will prefer more acidic water, and fish that like harder waters will prefer alkaline water. The dry, clay groundwater of Australia is acidic, meaning Australian rainbows prefer a pH range around 6.5. African cichlids prefer a more alkaline range of 8-8.5. However, many fish exist very comfortably in the 6-8 range. A stable environment is as important to the fish as any suggested pH range. Rather than chasing an ideal number, focus on keeping the pH levels consistent. However, it’s worth noting that most fish from acidic or alkaline waters will not breed unless they are in waters matching their native parameters.
How to adjust pH in your aquarium
To lower pH, like with all other parameters, your best bet is to start with reverse osmosis water. RO water is naturally slightly acidic, and because it lacks any KH, it can drop the pH levels even further. You can also introduce peat substrate or bogwood to your aquarium. These will release tannins into your tank that will increase the water’s acidity. Similarly, Indian Almond leaves when decomposing will release tannins into your tank.
To increase your aquarium’s pH, introduce calcium carbonate. Crushed coral, dolomite chippings and aragonite will all slightly boost your water’s pH. Limestone can also be added, which will elevate both pH and general hardness. Some aquarists add small amounts of baking soda, which will also increase the KH of the water. There are also chemical pH buffers widely available that can provide detailed directions on how much to add to see a desired increase. However, whenever you are adjusting pH (be it up or down), exercise extreme caution. As stated above, if the pH drops too low or swings too far upward, it has a detrimental effect on the health of your fish, coral, and plants. You can also increase aeration in the aquarium via aquarium pumps or air stones to help dissipate the carbon dioxide.
Learn more: What Is an Aquarium Air Pump and Is It Necessary?
Can I use tap water in my aquarium?It is possible to fill a tank with ordinary tap water, however, you will need to ensure that all traces of chlorine have been stripped from the water. Chlorine is toxic to aquatic life and will quickly kill your fish and poison your plants. Municipal water supplies use chlorine extensively to disinfect drinking water and eliminate waterborne viruses, protozoa, and bacteria. Chlorine produces a byproduct called hypochlorous acid. Through oxidation, chlorine can effectively destroy these pathogenic microorganisms and keep your tap water safe. However, the same chemical that neutralizes bacteria for humans is deadly to your fish. Exposure to chlorine causes fish to undergo necrosis, damaging their skin and gills and initiating serious respiratory problems. As they continue to breathe in the chlorine, they will suffer total organ failure and perish. Chlorine toxicity is a common mistake many novice fish-owners will make, but it is an avoidable misfortune.
However, truthfully, we have little control over the quality of our tap water without employing water filtration. While municipalities regulate city water to ensure it is safe for consumption, trace contaminants are still present. Mineral and metal content varies wildly from region to region. While Northeastern states like Maine and Vermont have slightly acidic, very soft water, the American Midwest’s water is very hard. Agricultural runoff, industrial waste, pharmaceuticals, microplastics, pesticides, and volatile organic compounds (VOCs) all make their way into city water. Though they may not be at levels that pose a threat to human health, fish are far more delicate and are very sensitive to their aqueous environment.
Learn more about aquariums: How to Maintain a Freshwater Aquarium | What Is an Aquarium Air Pump? | What Is an Aquarium Water Pump?
If you have any additional questions about reverse osmosis, please do not hesitate to contact us.





Comments
Question: can I run hot water through an R/O system?
Can you use softened water (using potassium chloride) before the RODI to provide suitable water for a fish only Marine aquarium?
Thanks very. It was very educating. Good job.